 
What is the Atomic Theory Timeline Project? So, how do you chart this scientific saga? What key milestones should your timeline include? Most importantly, how to create a project timeline as individuals? Let's explore these questions together, unraveling the fascinating journey of atomic models in science. It's about tracing the progression of atomic theory, from the earliest philosophical musings to the sophisticated quantum models of today. These probabilities form a region of space around the nucleus that he called orbitals (the most probable location of the electrons).Embarking on an Atomic Model timeline project can be likened to stepping into a time machine, journeying through the pivotal moments of scientific evolution.Stated that one could only predict the probability of where an electron could be (based on a mathematical formula), not its exact location.Discovered mass and electric charge of the electron by using the “Falling Drop” Method.Electrons can sometimes jump from one orbit to another by gaining or releasing energy.Electrons exist in orbits around the nucleus.Electrons are found outside of the nucleus.Nucleus contains most of the mass of an atom. 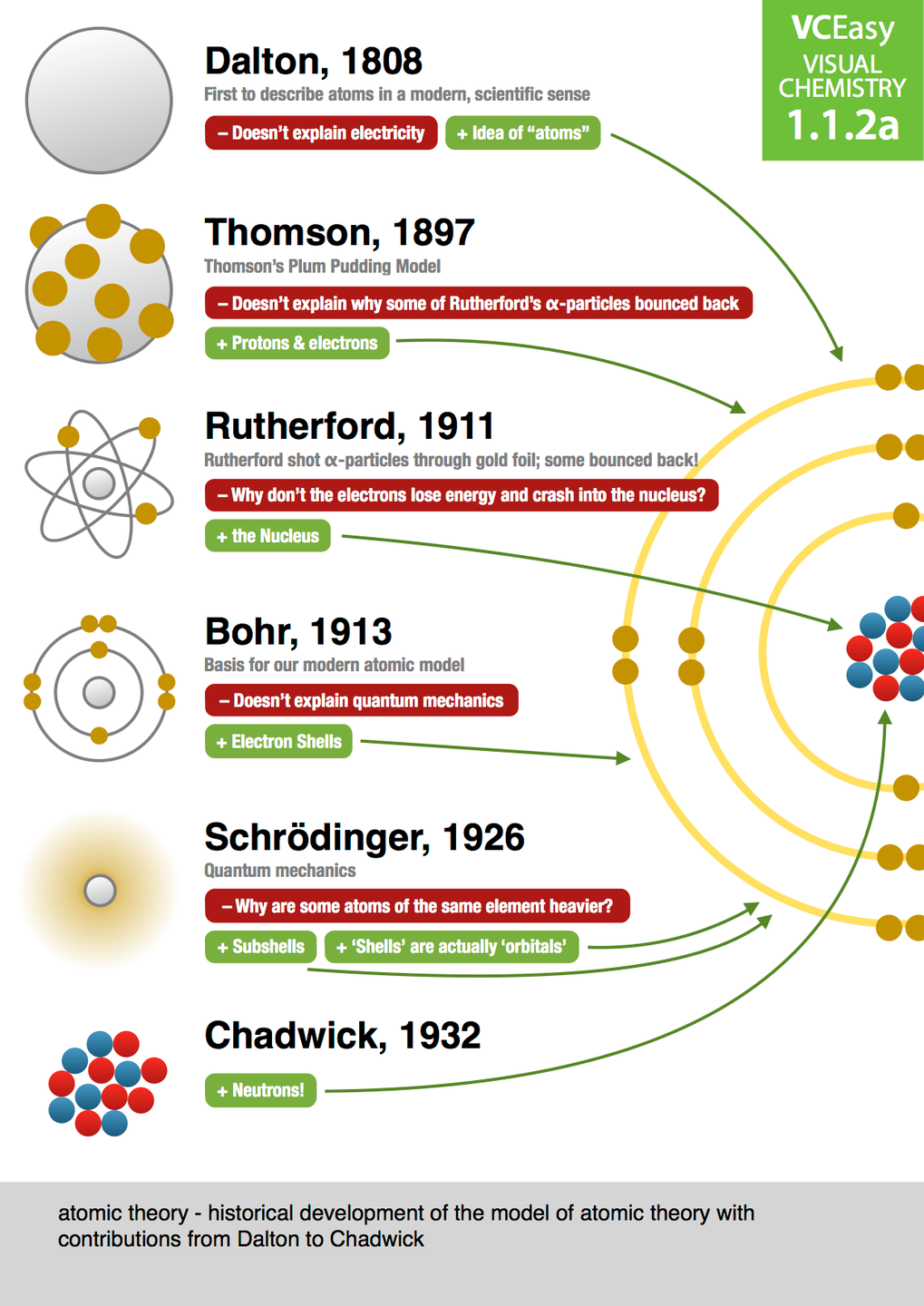
Atoms have a tiny dense center called ‘the nucleus’.Rutherford’s Contribution to Atomic Theory Most went straight through, some deflected at an angle, and a small percentage bounced straight back.Consisted of shooting alpha particles (positive charge) at a very thin piece of gold.His model shows negative charges as plums and positive charges as the pudding.He realized that neutral atoms must have equal amounts of positive and negative charge.Thomson’s Atomic Model“Plum Pudding” Model Beam will bend towards a positive charge, therefore, it must be made of negatively charged particles.Discovered electrons using the Cathode Ray Tube (CRT).If they fall apart, the must not be the smallest piece of matter out there.Her research indicated that some atoms are unstable and will disintegrate over time on their own.Discovered two new elements: polonium and radium in 1898.Discovered that a magnetic field could block radiation.When he placed metal between the uranium and photographic plates, the metal blocked the radiation.This proved that radiation could occur without outside energy such as the sun.When Becquerel placed uranium salt in a desk drawer with a photographic plate, radiation from the uranium formed an image on the plate.When elements react, their atoms combine in simple, whole-number ratios.Each atom of the same element is identical.Atoms are indestructible (can’t be divided into smaller parts).Thought matter was made of air, water, fire, and Earth.Thought atoms varied in size, shape and weight.Thought that all things were made of tiny, invisible, indestructible particles, called atoms.Explain the contributions of the following scientists to the development of the Atomic Theory: Democritus, Crookes, Becquerel, Curie, Millikan, Chadwickĭemocritus’s Theory(Foundation of Atomic Theory).4.1.3) Describe the experiments performed by Thomson and Rutherford and what the observations suggested about the structure of the atom.4.1.2) Explain the atomic models of the following scientists: Dalton, Thomson, Rutherford, Bohr, Schrödinger.4.1.1) List the main points of Dalton’s Atomic Theory and describe which parts are still believed to be true.Atomic History Timeline Major Contributors Main Discoveries
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
